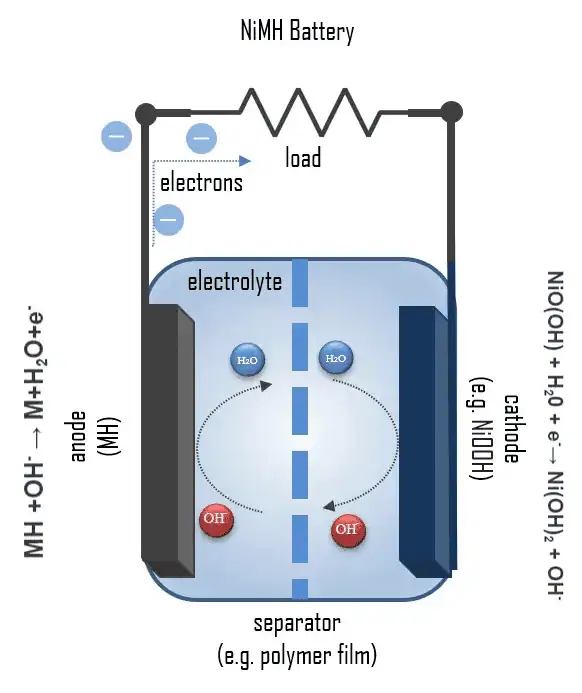
Batteries commonly have two electrodes: anode and cathode. Ions travel through the separator to either electrode during the charge and discharge cycles and release energy in the process.
Battery test cells can be built to include a third electrode. This is known as the reference electrode (RE). The RE allows for greater analysis of battery performance as it decouples test results between anode and cathode.
When researching battery materials, the use of a reference electrode (RE) allows researchers to measure and differentiate the contribution of each component of the cell to its overall performance. Three-electrode experiments help identify which electrode (anode or cathode) limits the cell performance during long-term testing. It is important to identify how each electrode is contributing to cell degradation under various test conditions instead of blindly experimenting with one or both.
Why is this important?
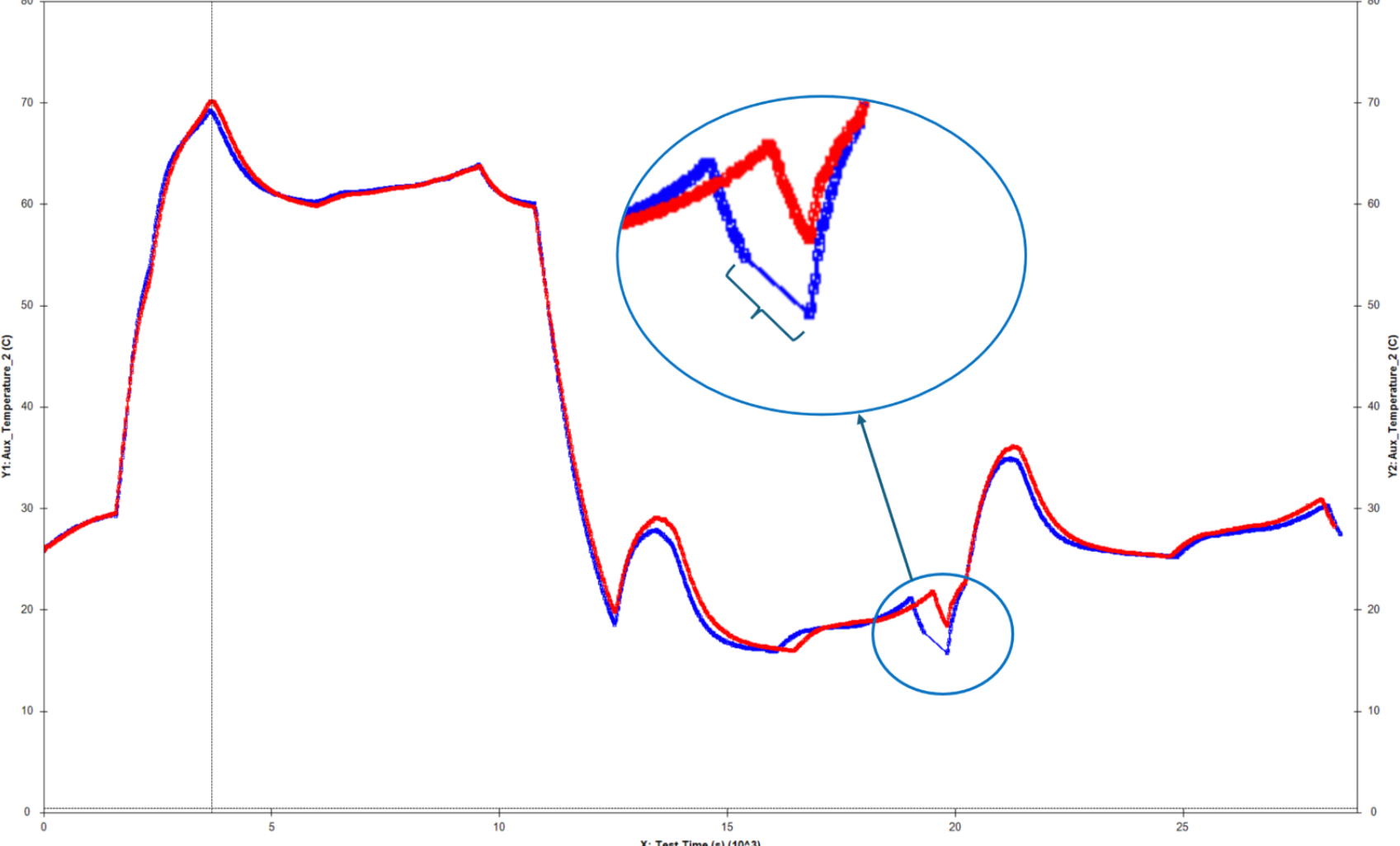
Most all electrochemical experiments and battery tests provide greater understanding of the cell when the anode and cathode results can be decoupled through use of a reference electrode. This extends to what are traditionally considered “industrial” applications as well. The dynamic charge-discharge profiles and fast charge simulations associated with commercial devices and electric vehicles can draw unique performance from a battery compared to low-rate constant current cycling.
Three-electrode testing is also beneficial for evaluating battery safety. Minter and Juarez-Robles highlight how fast-charging, which is a highly sought characteristic for electric vehicles, creates a great need to detect and monitor lithium plating occurring on a cell anode. [Minter RD, Juarez-Robles D, et al 2018 J Vis Exp., (135):57735.] This can best be achieved using a three-electrode cell during testing.
One fundamental goal of battery research is to develop cells that are long-lasting. This is especially important for electric vehicle and grid storage applications where the commercial cells and battery packs must last thousands of cycles and up to 10 years. Three-electrode testing allows researchers to identify the limiting factor in their cell to focus attention where improvement is needed most.
How the reference electrode is used in different testing situations
- During HPPC test, which are common for electric vehicle applications, the use of a reference electrode reveals electrode polarization.
- Performing EIS shows the decoupled impedance from anode and cathode individually when a three-electrode cell is utilized.
- The individual contribution of anode and cathode is revealed when demonstrating lithium loss due to SEI growth as a dominant aging mechanism.
- Differential capacity analysis can reveal changes in the voltage profile of anode and cathode and how they individually contribute to cell degradation.
The obstacles in creating a stable and reliable three-electrode cell
Comparing results from a new three-electrode experiment to other published results needs to keep as many variables consistent as possible, such as electrode size, material amount, cell uniformity, etc., or else attempt to normalize results. This is a principal reason why traditional cell types are modified to incorporate a reference electrode as “homemade” cell, so results are easier to compare with minimal normalization. Researchers wish to demonstrate and compare their results to existing two-electrode data of the same cell type (cylindrical, pouch, coin). However, since most battery material work is conducted using coincells, this is the natural choice for three-electrode experiments to compare the new results with the vast amount of tradition two-electrode data in publication. The new experimental data will decouple the anode and cathode and provide new insights.
Other commercial three-electrode cells such as Swagelok-style or split cell designs are costly and not practical to implement and scale, and can sometimes to complicated to build and use. Test results from these types of cells must also be normalized when comparing with traditional battery formats (coin, cylindrical, pouch, etc.).
Arbin’s three-electrode test cell configuration and its benefits
The novel “3E” coincell has the same surface area as a traditional CR2032 coincell and makes it ideal for comparing results across all published coincell data. It provides users with the ability to rapidly prototype new materials by performing large-scale three electrode testing. Traditional methods have proven too expensive and provided inconsistent results. Arbin’s new 3E Coin Cell provides users with an affordable, easy to use three-electrode cell holder that allows for long-term cycling, and provides consistent results between samples. The low unit cost, disposable design, and easy-to-build coin cell structure allows users to quickly build a large number of cells for materials research testing. The 3E Coin Cell interfaces with Arbin’s 3E Coin Cell Holder, and connects directly with our MSTAT product series
The alternative solutions for decoupling test results between anode and cathode described in sections above exist because this data has the ability to expedite battery research and development and bring new battery chemistries to market faster.
Arbin’s latest generation of LBT and MSTAT high-precision battery test equipment has attracted attention from both academic and industry researchers due to its ability to also expedite the battery development process. 24-bit resolution, extreme precision, and state-of-the-art thermal management that are standard with Arbin produce more detailed and consistent results than other battery testers. This has led multiple industry partners to team up with Arbin on collaborative projects including ARPA-E grants. General Motors has permitted Arbin to license and commercialize a new three-electrode cell design that can further enhance and accelerate their battery testing program.